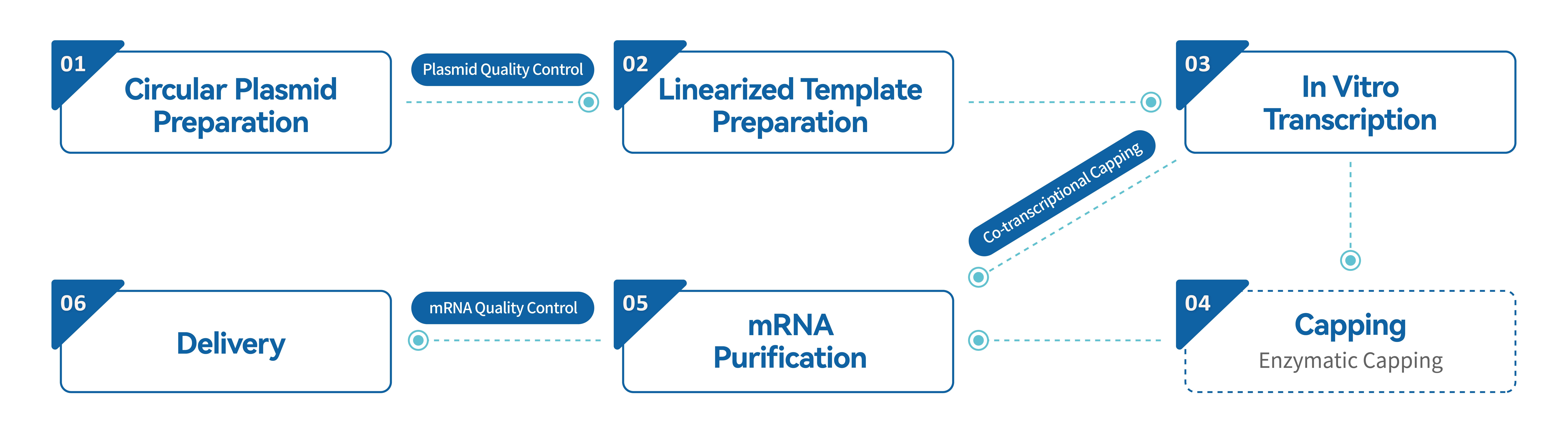
Hzymes Biotech possesses the necessary restriction endonucleases for the preparation of linearized plasmid templates, enzyme raw materials for in vitro transcription, modified nucleosides, cap analogues, purification, and quality control materials. It can provide customized synthesis of mRNA with high yield, purity, and cost-effectiveness in different specifications.
Hzymes Biotech provides full process services from sequence design to mRNA synthesis, supporting the synthesis of various forms of RNA such as linear mRNA, saRNA, CircRNA, etc. It also has a professional quality control platform, which can better safeguard the research and development of RNA related products for customers.
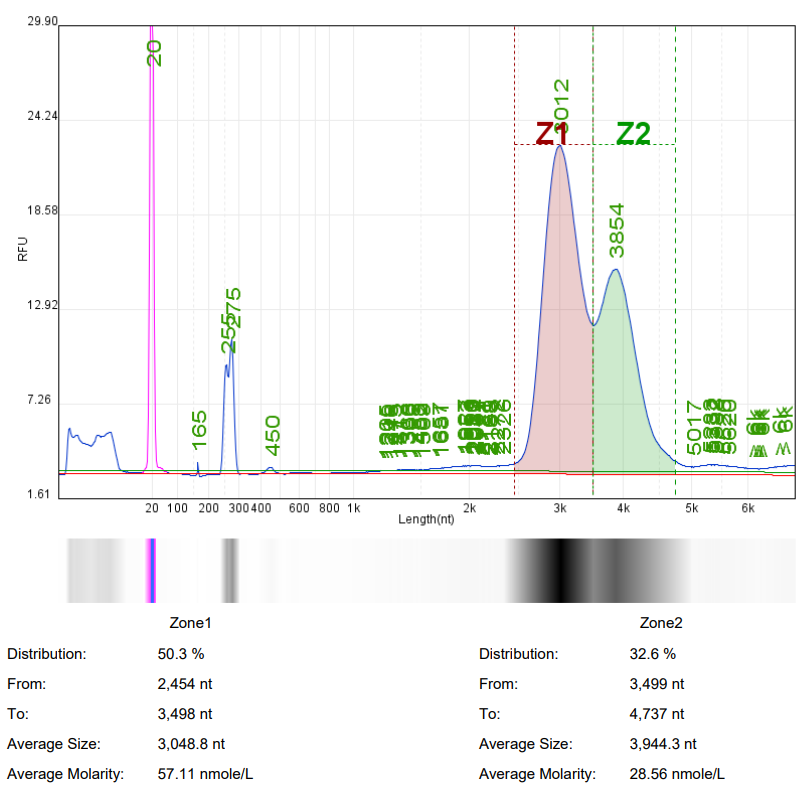
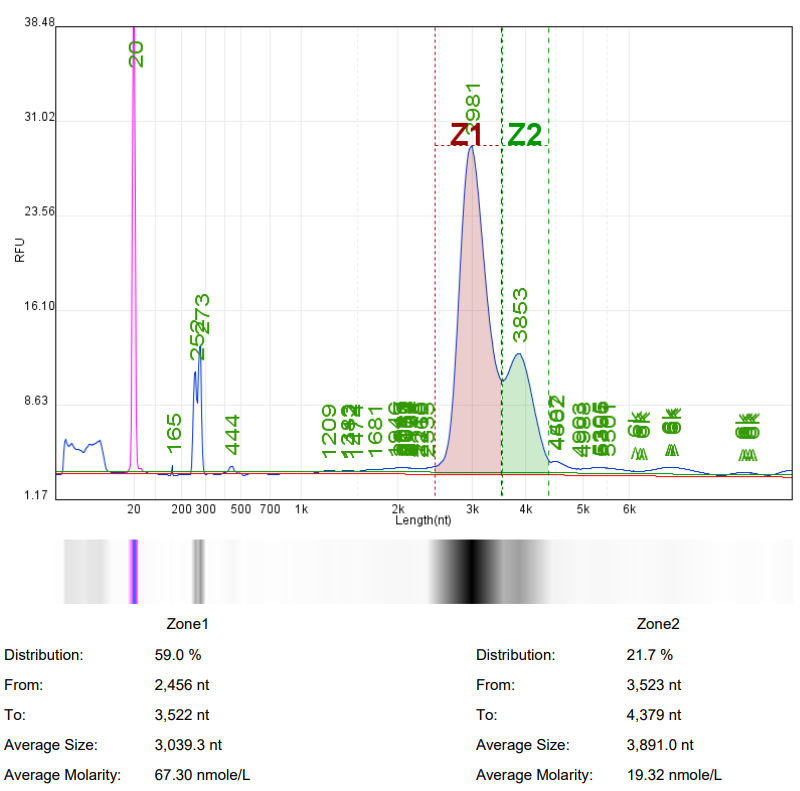
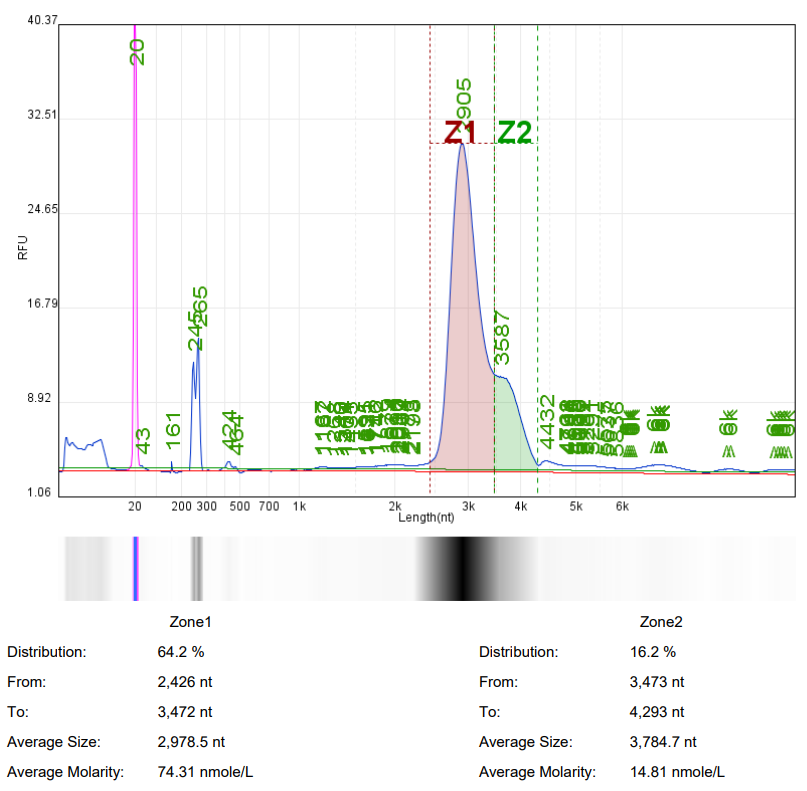
Self amplifying RNA (saRNA) is an mRNA that encodes a replicase and can self replicate within cells using itself as a template. SaRNA contains the basic elements of mRNA (cap, 5'UTR, 3'UTR, and variable length polyadenylation tail), with a large open reading frame (ORF) at the 5 'end encoding four non structural proteins (nsP1-4) and a subgenome promoter to facilitate RNA self replication. The self replication property of saRNA can achieve lower dosages of RNA drugs, prolong protein expression time in vivo, and reduce side effects.
Hzymes Biotech can provide customized synthesis services for saRNA, selecting the optimal expression elements, sequence design, and base modification to ensure efficient in vivo and in vitro expression of saRNA, and better assist in the research and development of saRNA products.
Case 1 | Case 2 | |
Product name | saRNA1 | saRNA2 |
Hat adding method | Cotranscription | Cotranscription |
Modified nucleoside | UTP | UTP |
mRNA length | 8000nt | 9020nt |
A260/280 | 2.1 | 2.1 |
Appearance | No impurities/precipitation | No impurities/precipitation |
Analysis of mRNA Integrity by mRNA Liquid Capillary Electrophoresis Nucleic Acid Analyzer | 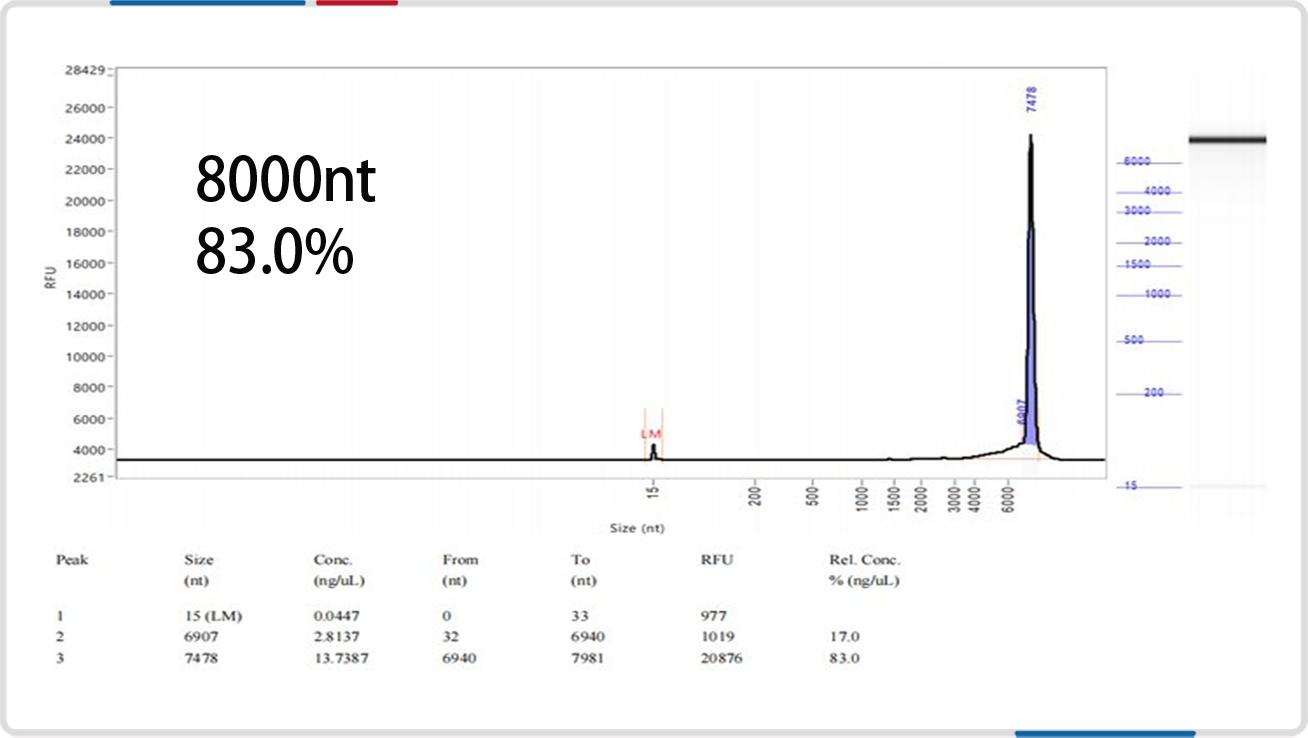 | 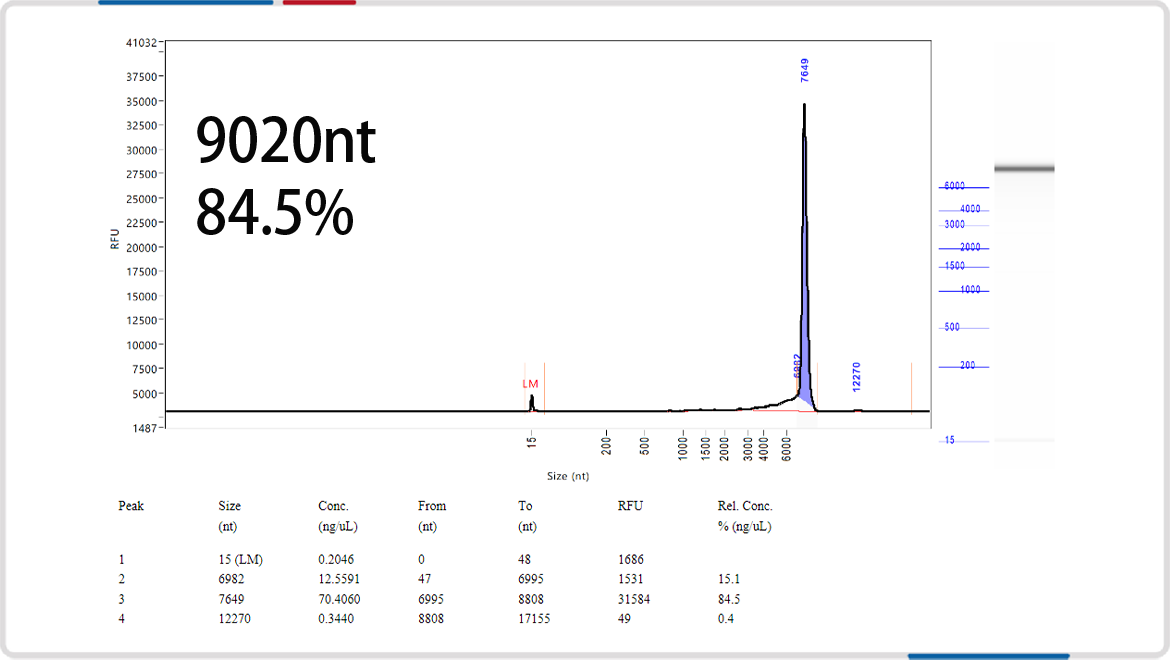 |

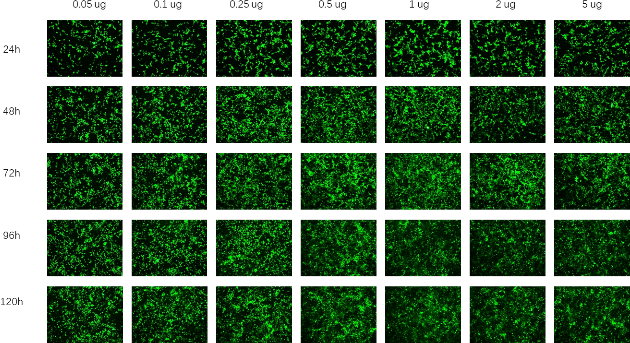
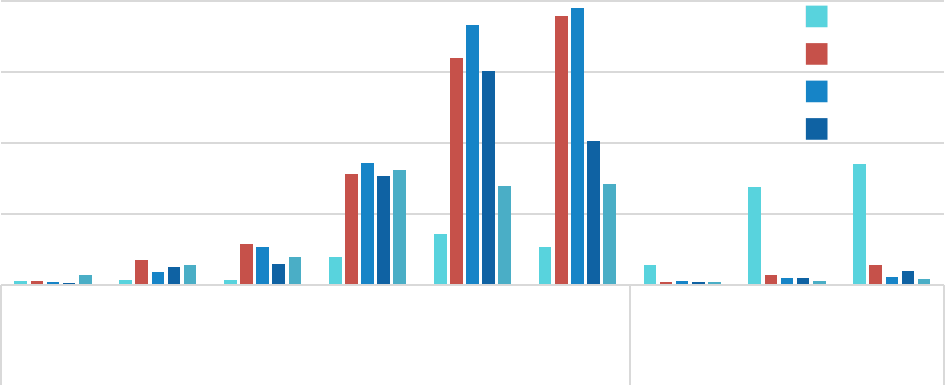
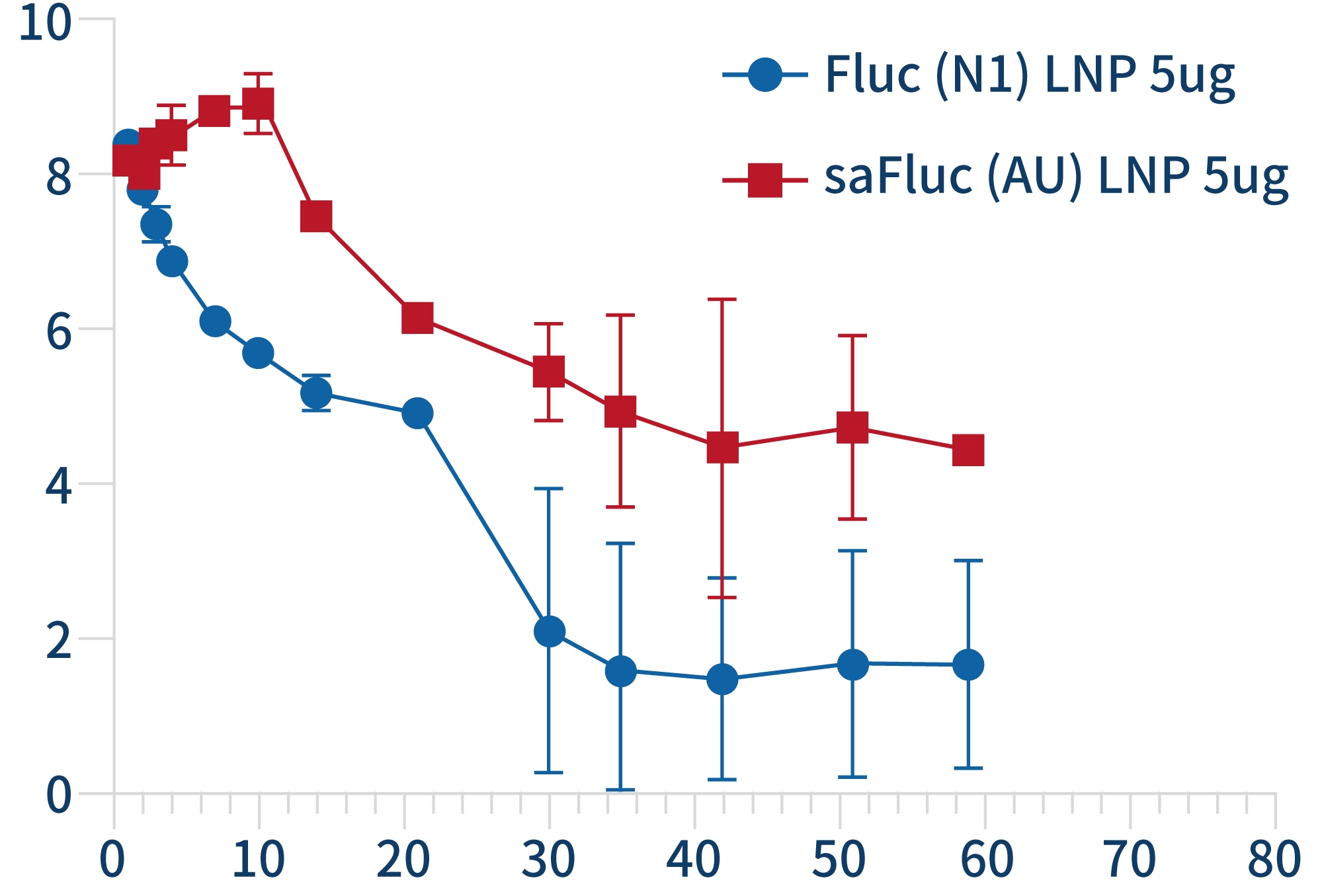

Circular RNA (circRNA) is a novel non coding RNA produced by reverse splicing of pre mRNA precursors. Unlike traditional linear RNA, circRNA has a closed circular structure formed by covalent bonds at the 5 'and 3' ends, which is not affected by RNA nucleases, resulting in more stable expression, less degradation, and lower immunogenicity.
Hzymes Biotech can provide customized synthesis services for CircRNAs, supporting the synthesis of circular RNAs of different fragment lengths, ensuring efficient expression of CircRNAs in vivo and in vitro, and better assisting in RNA therapies such as Car-T and protein replacement in vivo, as well as the research and development of RNA related products such as gene regulation and drug delivery.







Service category | Project | Duration (working days) | Deliver |
Sequence design (not undertaken separately) | Sequence design | 5 | Sequence file |
Templates preparation | Plasmid synthesis | 20 | Plasmid sequencing files, if requested by the customer, provide synthesized raw plasmids (1-2 μ g) |
Plasmid amplification | 5 | Plasmid sequencing files, if requested by the customer, can be provided to the customer if there are remaining plasmids produced, and no additional preparation will be made if there are no remaining plasmids | |
mRNA raw material synthesis | Synthesis of mRNA Raw Solution - Non nucleotide Modified mRNA (Research Grade) | 5 | mRNA stock solution and COA |
mRNA raw material synthesis - nucleotide modified mRNA (scientific grade) | 5 | ||
mRNA Raw Solution Synthesis - Non nucleotide Modified mRNA (Industrial Grade) | 5 | ||
mRNA raw material synthesis - nucleotide modified mRNA (industrial grade) | 5 |
Service category | Project | Duration (working days) | Test method |
Can provide quality inspection services (not separately undertaken) | Double-stranded RNA | 1 | ELISA |
Total protein residue | 1 | NanoOrange | |
DNA template residue | 1 | Fluorescence quantitative PCR | |
Endotoxin | 1 | TAL | |
RNase residue | 1 | Fluorescence probe method | |
DNase residue | 1 | Fluorescence probe method | |
Host DNA residue | 1 | Fluorescence quantitative PCR | |
NTP and hat like residue | 1 | HPLC |

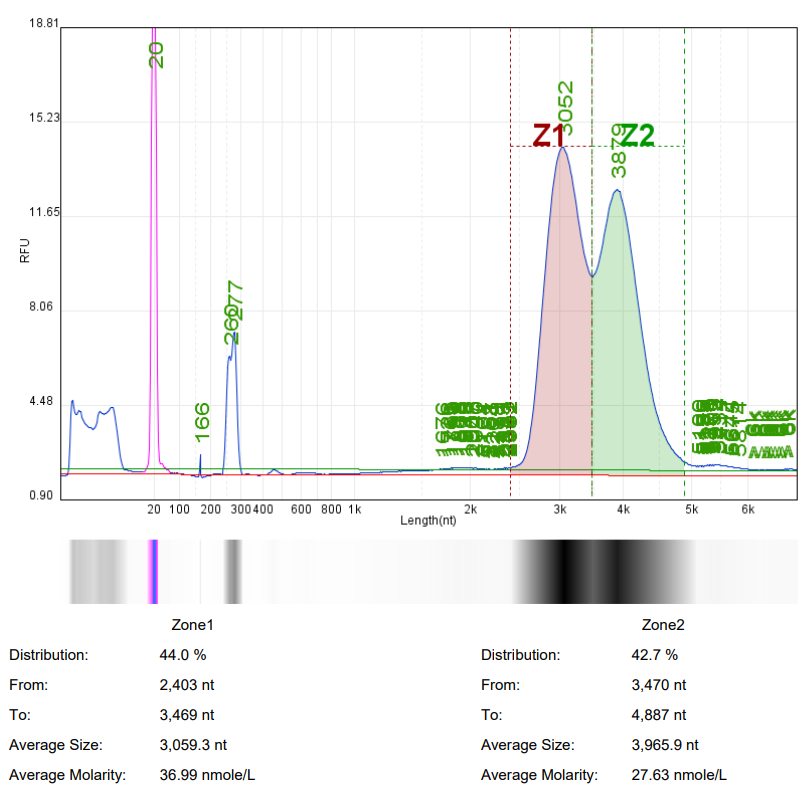
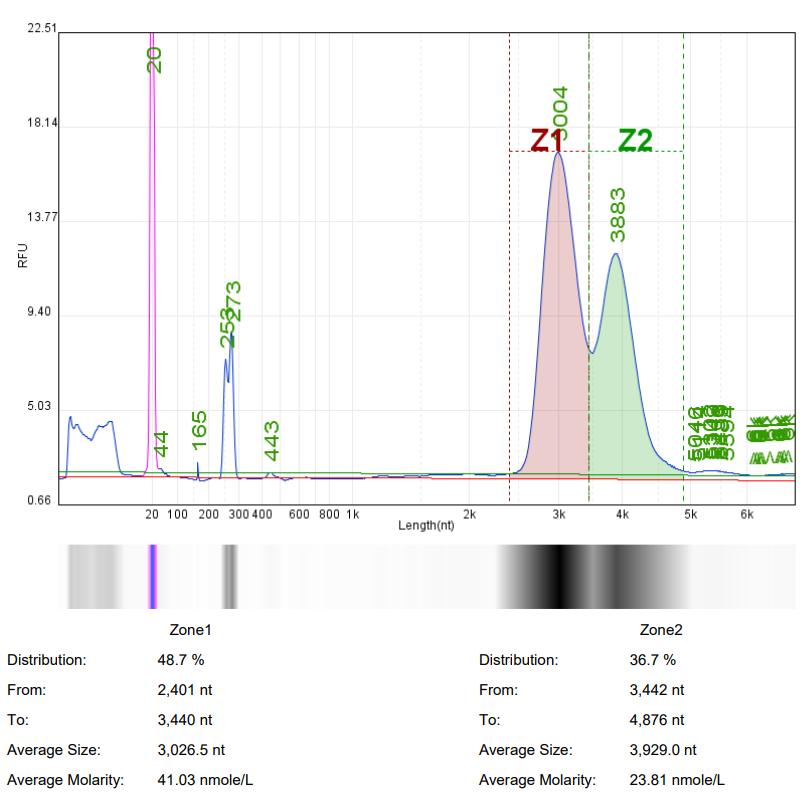
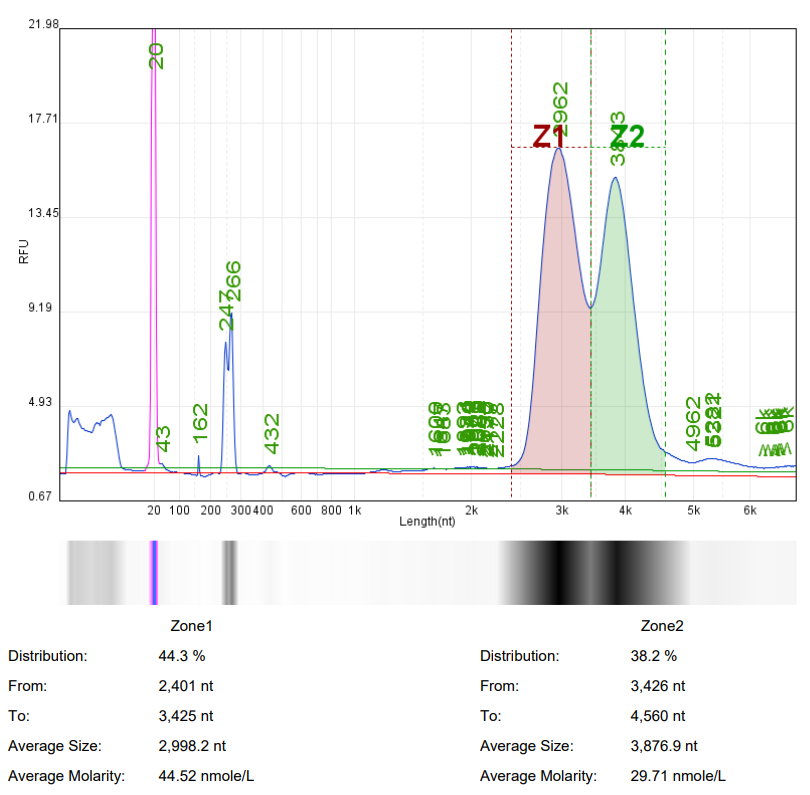


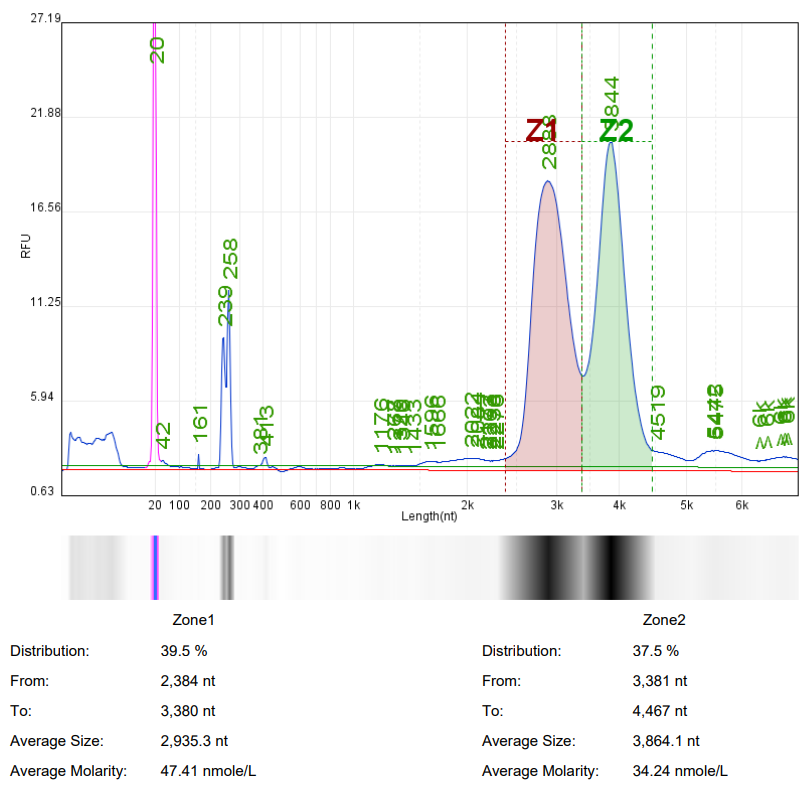



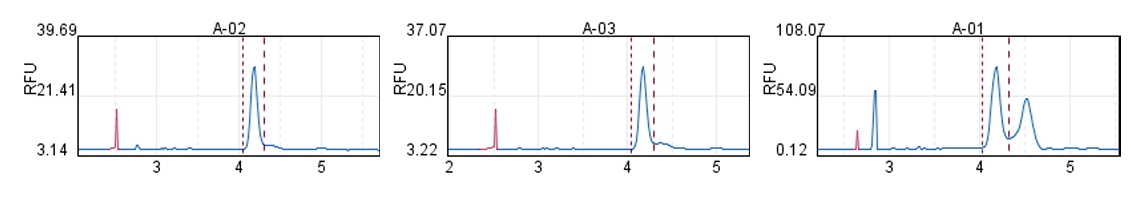
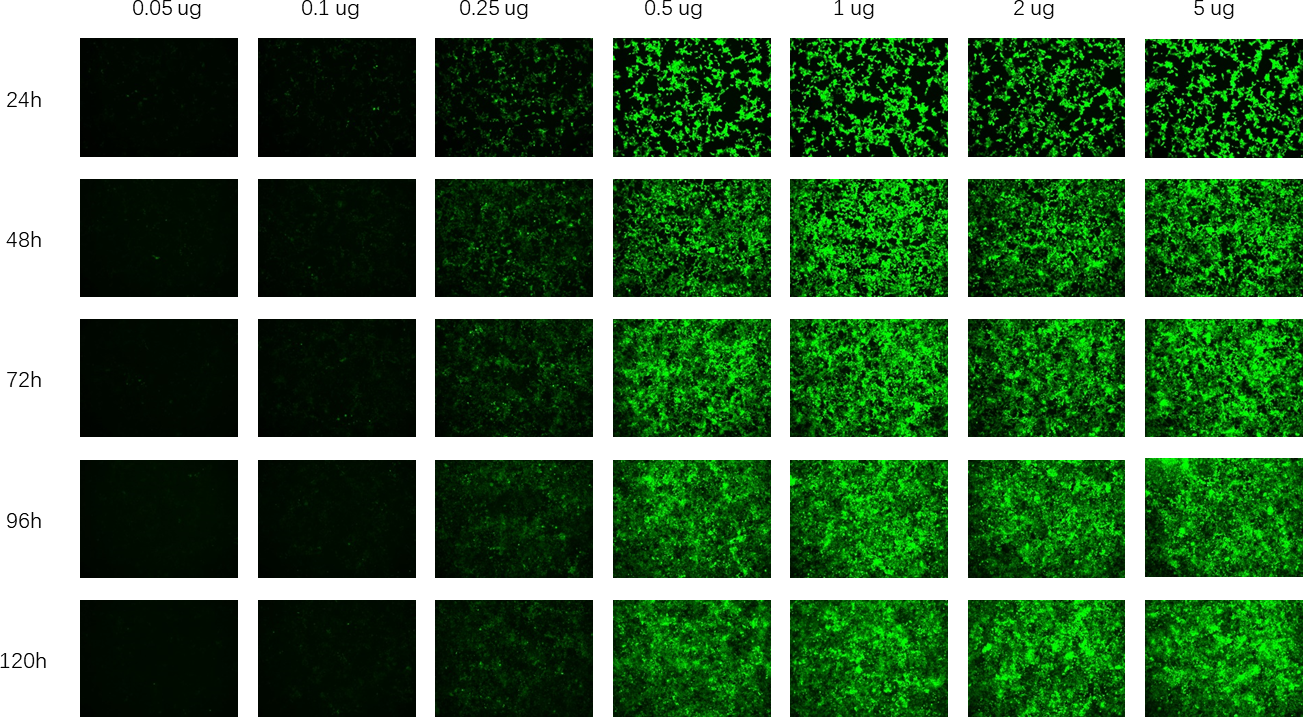
In mRNA drug research, it is often necessary to use ready-made mRNA raw materials as cell or animal experimental controls, or to explore purification processes, screen fillers, optimize mRNA expression levels, and evaluate carrier delivery efficiency.
Hzymes Biotech provides a variety of IVT mRNA raw materials in stock, including COVID-19, EGPF encoding, Fluc or mCherry reporter genes, DNA endonuclease gene Cas9, erythropoietin gene EPO precursor, etc. Linear mRNA and saRNA forms can be selected to provide more assistance for mRNA research.
Case 1 | Case2 | Case 3 | Case 4 | |
Product name | mRNA1 | mRNA2 | mRNA3 | saRNA |
Hat adding method | Co transcription method with cap | Co transcription method with cap | Co transcription method with cap | Co transcription method with cap |
Modified nucleoside | N1-Me-pUTP | N1-Me-pUTP | N1-Me-pUTP | N1-Me-pUTP |
mRNA length | 500nt | 1000nt | 2000nt | 4000nt |
A260/280 | 1.85 | 1.84 | 1.88 | 1.92 |
Appearance | No impurities/precipitation | No impurities/precipitation | No impurities/precipitation | No impurities/precipitation |
Capillary electrophoresis nucleic acid analysis mRNA integrity | 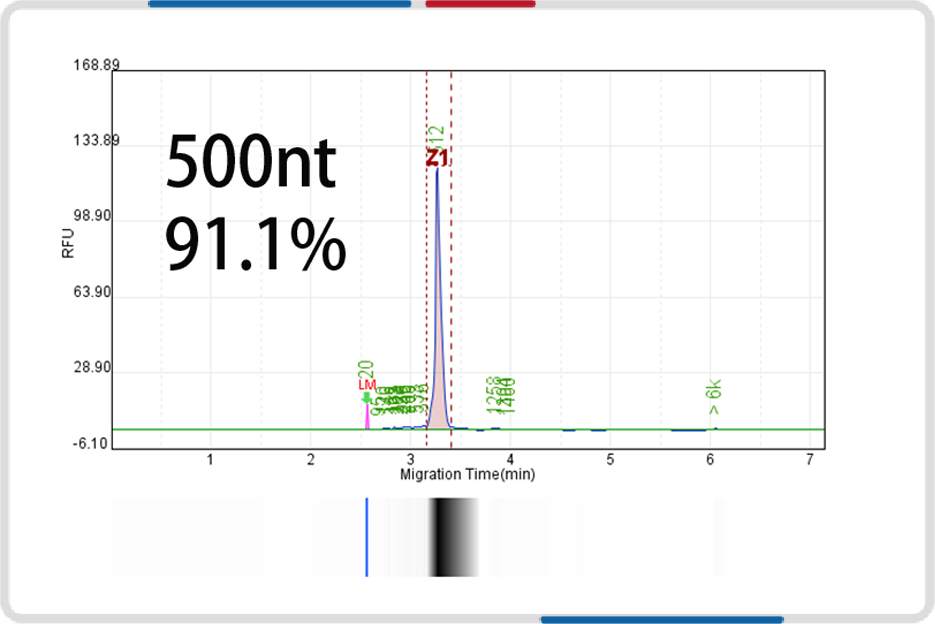 | 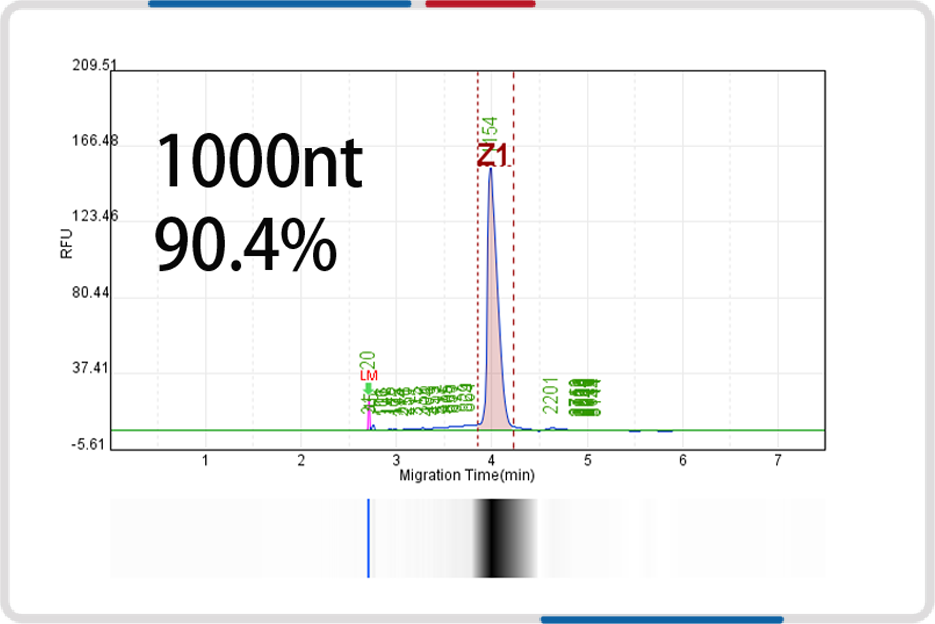 | 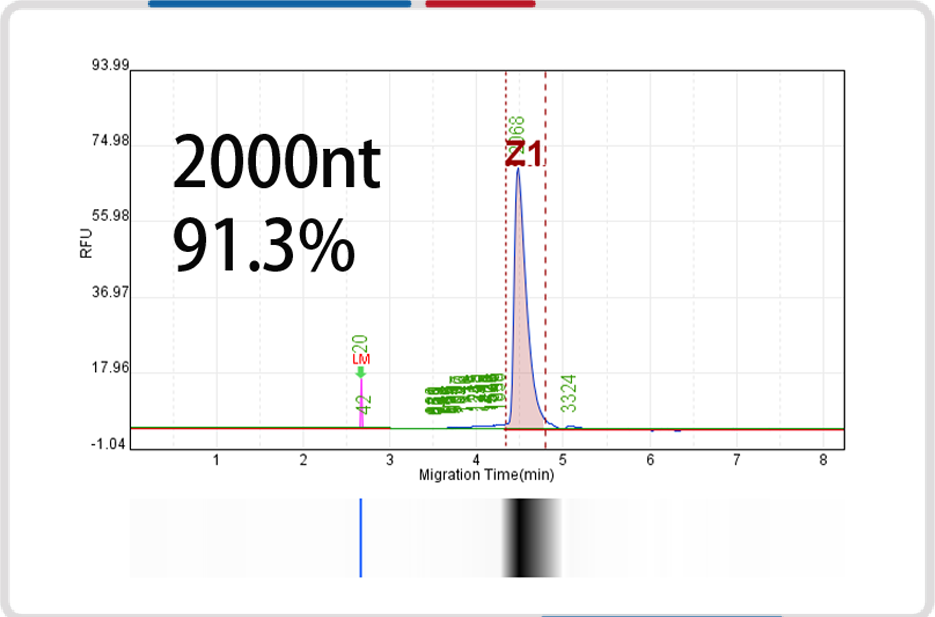 | 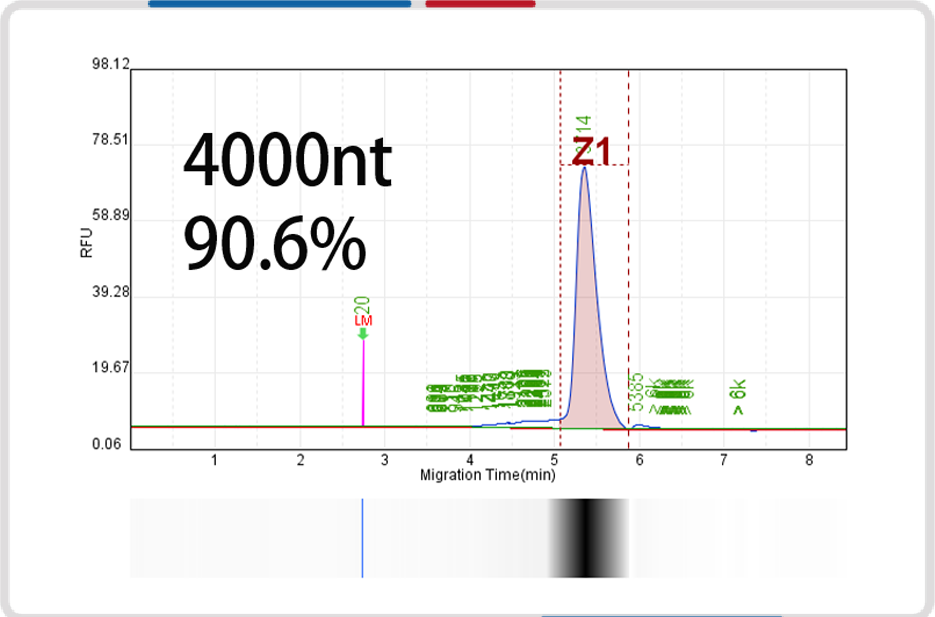 |
Testing items | Case 1 | Case 2 | Case 3 | Case 4 |
mRNA integrity | 90.6% | 91.9% | 93.2% | 91.4% |
Residual endotoxins | <10EU/mg | <10EU/mg | <10EU/mg | <10EU/mg |
RNase residue | <1.56x10^-9 U/μL | <1.56x10^-9 U/μL | <1.56x10^-9 U/μL | <1.56x10^-9 U/μL |
Protein residue | <0.5μg/mg | 0.521μg/mg | 0.53μg/mg | 0.5μg/mg |
Residual plasmid template | 3.07x10^-4 μg/mg | 5.18x10^-6 μg/mg | 1.05x10^-4 μg/mg | 7.21x10^-5 μg/mg |
dSRNA residue | 0.0789% | 0.1749% | 0.0389% | 0.3266% |
Cap rate | 98.71% | 100% | 98.77% | 100% |
Poly (A) distribution | Qualified | Qualified | Qualified | Qualified |
Sequence correctness | Qualified | Qualified | Qualified | Qualified |


