T7 RNA polymerase, as the most important raw material in IVT, plays a decisive role in mRNA quality. Hzymes Biotech has five major technology platform based on enzyme gene resource mining, enzyme directed evolution, enzyme artificial intelligence design, enzyme large-scale manufacturing, and application technology research and development of enzyme catalytic systems. It can provide a complete set of materials for mRNA synthesis, as well as multiple T7 RNA polymerization mutants. Based on in-depth research on different templates, mutants, and IVT systems, it has screened and established its own IVT Buffer library to meet various production needs and application scenarios of customers.
mRNA detection service
Quality control, as the most important part of drug development, is of paramount importance in the analysis of drug components, impurities, and other factors during the drug development process. Hzymes Biotech provides mRNA drug quality inspection services
Quality inspection items | Quality inspection method |
| Concentration | Nanodrop method |
| Concentration | Nanodrop method |
ELISA | |
integrity | CE |
SEC-HPLC | |
dsRNA | ELISA |
DNA template residue | qPCR |
Capping efficiency | LC-MS |
Poly (A) tail length | LC-MS |
Product related impurities - polymer quantification | SEC-HPLC |

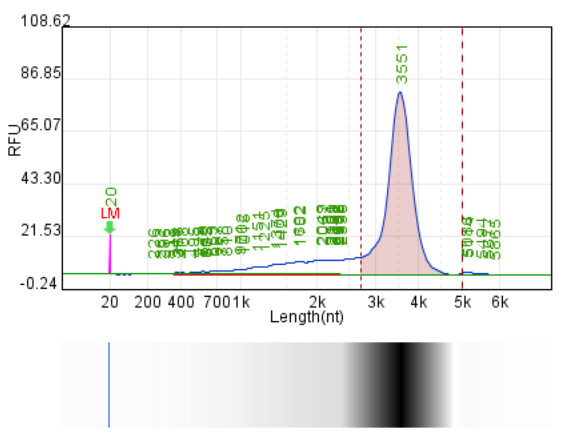



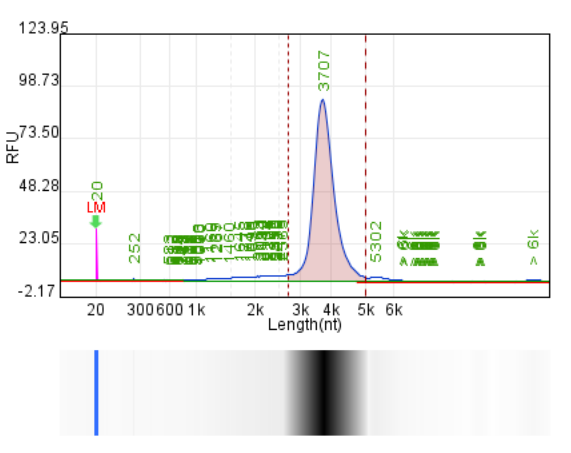

System | Integrity | Production | Label dsRNA content |
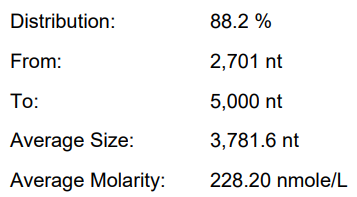
Original system | 77.70% | 146.4μg/20μLReaction system | 2.35% |
Hzymes optimizes IVT system 1 | 87.40% | 214μg/20μLReaction system | 1.02% |
Hzymes optimizes IVT system 2 | 85.90% | 225.1μg/20μLReaction system | 0.16% |




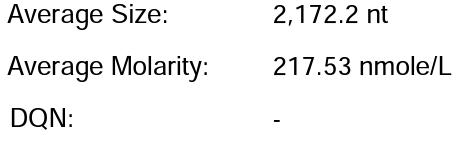

System | Integrity | Production |
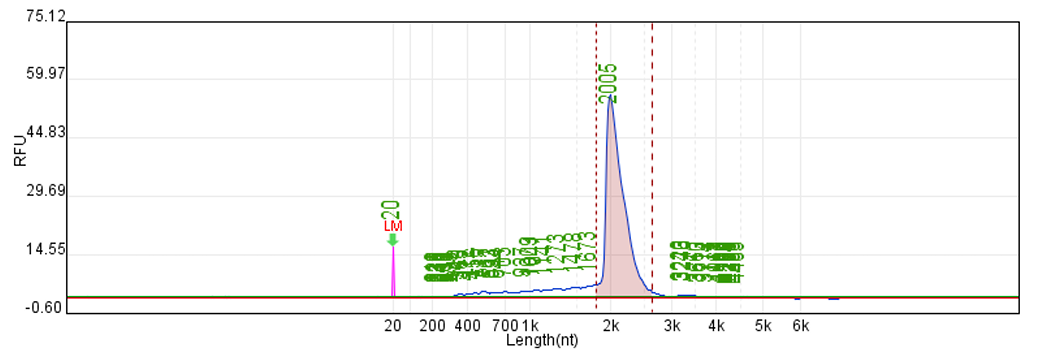
Customer system | No clear main strip | 76μg/20μLReaction system |
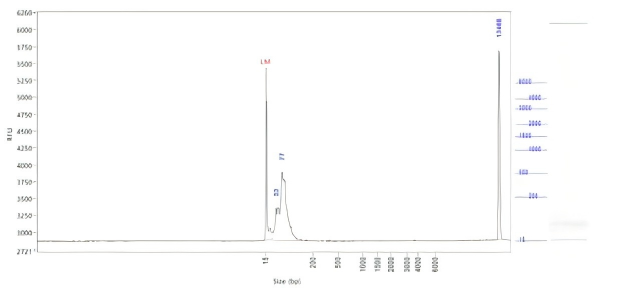
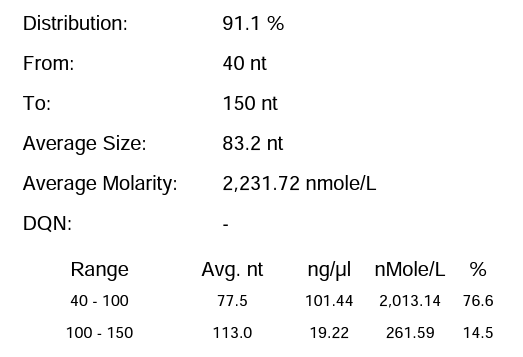
Hzymes optimizes IVT system 1 | 91.10% | 197.64μg/20μLReaction system |
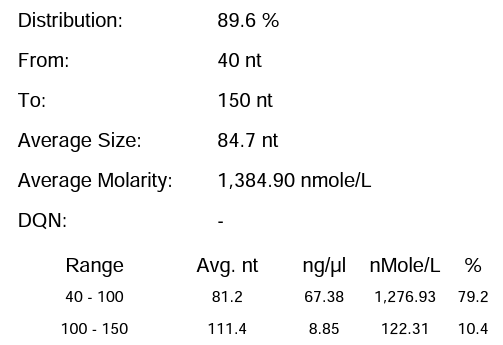
Hzymes optimizes IVT system 2 | 89.60% | 212.7μg/20μLReaction system |
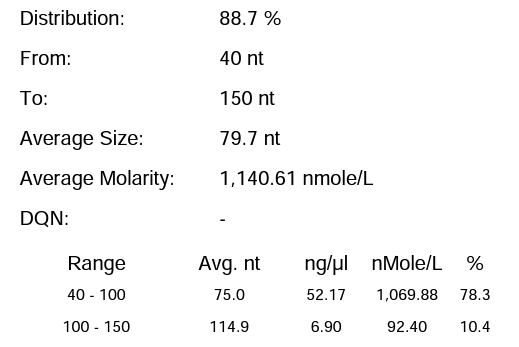
Hzymes optimizes IVT system 3 | 88.70% | 210.39μg/20μLReaction system |
Hzymes optimizes IVT system 4 | 92.60% | 212.54μg/20μLReaction system |
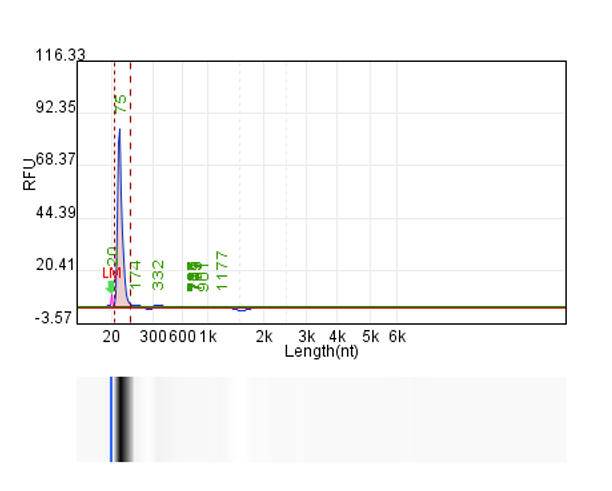
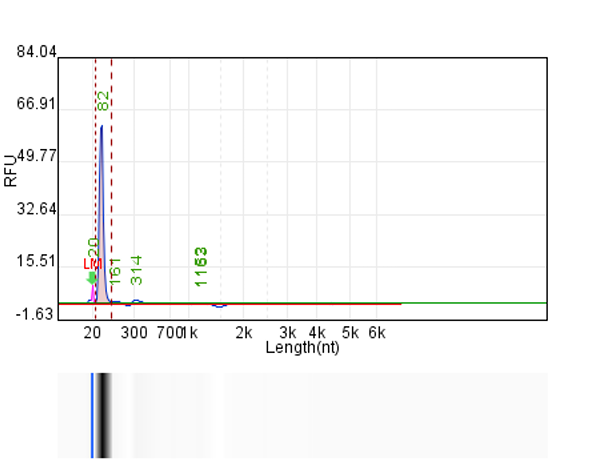
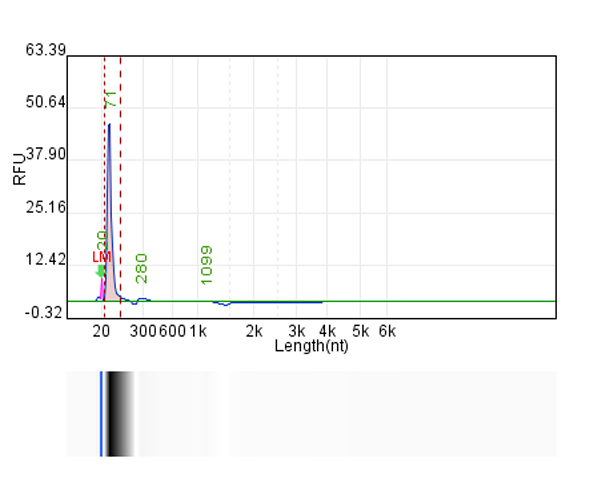
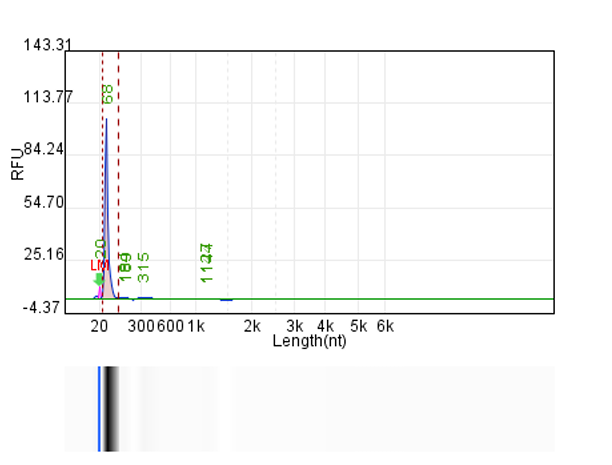
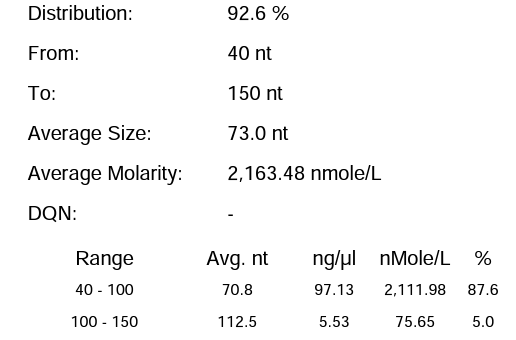

System | Integrity | Production |
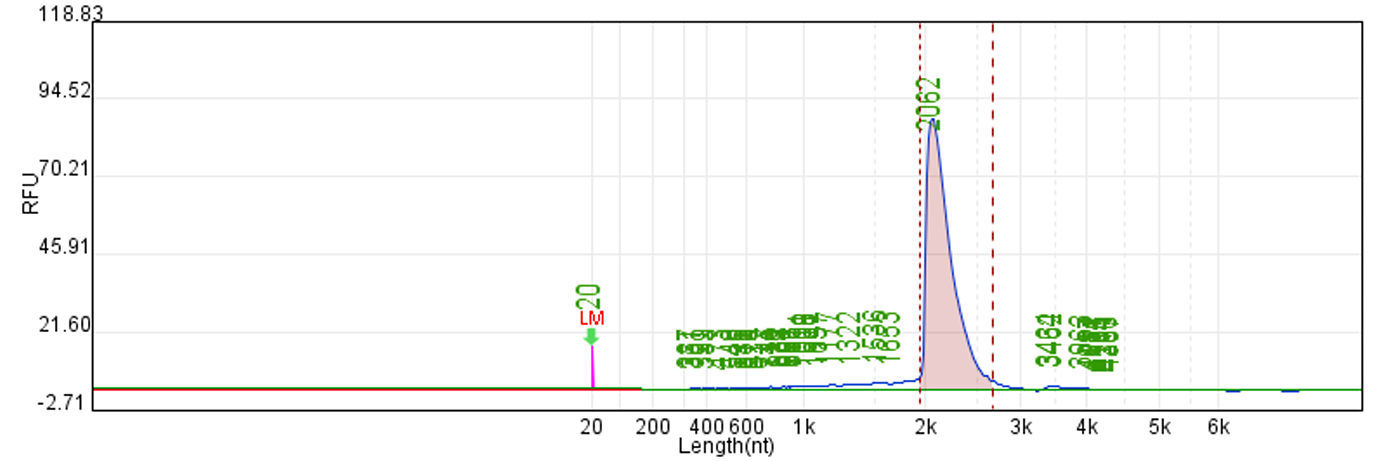
Customer system | 88.67% | 222μg/20μLReaction system |
Hzymes optimizes IVT system 1 | 90.24% | 228.5μg/20μLReaction system |
Hzymes optimizes IVT system 2 | 91.82% | 231.4μg/20μLReaction system |
Hzymes optimizes IVT system 3 | 91.63% | 233.3μg/20μLReaction system |
Hzymes optimizes IVT system 4 | 89.04% | 215.7μg/20μLReaction system |
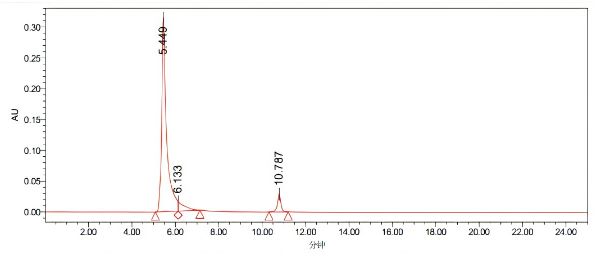
Processing channel: PDA260.0 nanometers (PDA200.0 to 400.0 nanometers (at 1.2 nanometers))
Processing channel | Retention time (minutes) | Area | %Area | Peak height | |
1 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 5.440 | 4060130 | 88.67 | 31420 |
2 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 6.133 | 318002 | 5.68 | 15482 |
3 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 10.787 | 316960 | 5.66 | 28881 |
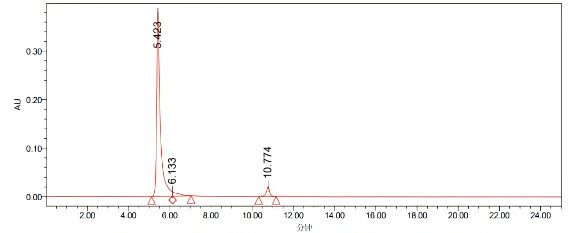
Processing channel: PDA260.0 nanometers (PDA200.0 to 400.0 nanometers (at 1.2 nanometers))
Processing channel | Retention time (minutes) | Area | %Area | Peak height | |
1 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 5.423 | 4725664 | 91.82 | 380268 |
2 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 6.133 | 181471 | 3.53 | 8910 |
3 | PDA 200.0 nanometers (PDA 200.0 to 400.0 nanometers (at 1.2 nanometers)) | 10.744 | 239565 | 4.65 | 20556 |
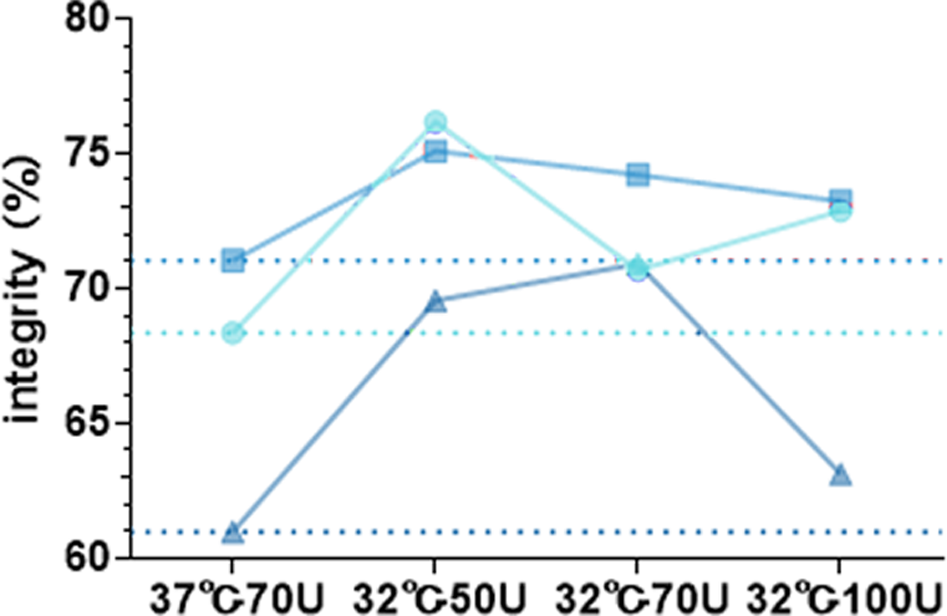
Temperature | Enzyme | Enzyme quantity | mRNA 1(9016nt) | mRNA 2(8497nt) | mRNA 3(10255nt) | |||
Production(μg) | Integrity | Production(μg) | Integrity | Production(μg) | Integrity | |||
32℃ | M6 | 50U | 63.35 | 76.16% | 74.95 | 75.08% | 92.7 | 69.55% |
70U | 85.8 | 70.67% | 106.7 | 74.20% | 108.6 | 70.91% | ||
100U | 108.55 | 72.87% | 130.8 | 73.22% | 122 | 63.12% | ||
37℃ | M6 | 70U | 200.15 | 68.34% | 219.85 | 71.02% | 213.05 | 60.97% |



Taking 4000nt mRNA as an example, the purification process related data are as follows:
Step | Cycle No. | Load | Elution | |||||||||
CE (%) | SEC_HPLC (H/M/L)(%) | Res.Protein (ug/mg) | Yield(%) | CE(%) | SEC_HPLC (H/M/L)(%) | Res.Protein (ug/mg) | dsRNA (ug/mg) | HCD (ug/mg) | Res.DNAtemplate (ug/mg) | Endotoxin (EU/mg) | ||
TFF 1 | Cycle 1 | 76 | - | - | 100 | 78.8 | ND/97.3/2.8 | 8.5 | 0.69 | ND | 2.07×10-15 | <9.4 |
AC | Cycle 1 | 71.5 | ND/97.9/2.1 | 6.3 | 92.1 | 82.5 | 3.7/95.1/1.2 | 0.2 | 0.60 | ND | 1.04×10-15 | <17.5 |
Cycle 2 | 76.7 | ND/96.9/3.1 | 6.3 | 94.0 | 83.0 | 0.8/98.0/1.2 | 0.2 | 0.94 | ND | 9.36×10-16 | <17.2 | |
TFF 2 | Cycle 1 | 82.8 | 3.6/95.4/0.9 | 0.2 | 99.6 | 82.5 | ND/99.6/0.4 | / | 0.68 | / | / | <6.8 |




General purification process and function of downstream technology for Hzymes Biotech mRNA
·Step 1 UF/DF filtration: Concentrate the IVT reaction product to the target volume and replace it with buffer solution through washing filtration, reducing RNA polymerase, DNA template NTPs、 Impurities such as cap enzymes, reagents, inhibitors, etc.
·Step 2: Capture mRNA: Use oligo dT filler to capture mRNA containing PolyA tail and remove impurities such as free nucleotides, short chain transcripts, enzymes, and other IVT reaction components.
·Step 3 UF/DF concentration and liquid exchange: Use UF/DF for concentration and liquid exchange to replace the target product into the final formulation buffer.
·Step 4: Sterilization filtration: Perform 0.22 µ m membrane sterilization filtration.
Finally, these purified mRNA drug substrates can be encapsulated into LNP for the next step.
Purification process development and data display
In the field of mRNA, in addition to designing optimized mRNA sequences and developing efficient and safe delivery vectors, developing simple, fast, large-scale, and economically efficient mRNA preparation processes is also one of the most important innovations in mRNA technology. In the production process of mRNA, purification after IVT is crucial for the safety and efficacy of the final mRNA product, as the content of impurities can affect the translation efficiency of mRNA and alter immunogenicity. Therefore, it is necessary to remove impurities, including residual substrates dsRNA、Abortive mRNA, DNA templates, protease residues, etc. Common purification methods include ultrafiltration, chromatography, precipitation, etc.











mRNA purification quality inspection items
Quality inspection items | Quality inspection method |
| Concentration | Nanodrop |
ELISA | |
Integrity | CE |
SEC-HPLC | |
DNA template residue | qPCR |
Capping efficiency | LC-MS |
Poly (A) tail length | LC-MS |
| Total protein residue | Nanoorange |
Product related impurities - NTP residue | SEC-HPLC |
| Product related impurities - polymer quantification | SEC-HPLC |
| Product related impurities - residual T7RNA polymerase content | ELISA |
| Endotoxin | TAL |
RUN | Test | Load | UFDF pool filter |
1 | Entrapment efficiency(%) | 97.66 | 94.84 |
Particle size(nm) | 67.37 | 71.51 | |
PDI | 0.09 | 0.12 | |
2 | Entrapment efficiency(%) | 95.69 | 95.00 |
Particle size(nm) | 66.90 | 73.94 | |
PDI | 0.13 | 0.16 |




The foundation of the packaging/loading process is the design and development of the delivery system. A well-designed delivery system is necessary to prevent mRNA molecules from being degraded by RNase after entering the human body, effectively deliver them to the target site, cross the cell membrane, and release them inside the cell. Hzymes adopts the current mainstream lipid nanoparticle delivery system.
The key to the preparation of mRNA LNP encapsulation lies not only in the formulation of lipid components, but also in the control of the process, that is, how to control the contact and interaction between mRNA and lipid components to form stable, uniform, and high-yield mRNA LNP complexes. Hzymes also adopts the current mainstream microfluidic mixing technology. Due to the solubility of mRNA in slightly acidic aqueous phase and liposomes in ethanol, the mRNA solution and liposome solution are mixed under high pressure to form two opposing jets. Intense turbulence ensures thorough mixing of all components, while the ethanol phase is diluted and the pH of the solution changes. Liposomes precipitate to form lipid nanoparticles and form encapsulated complexes with mRNA.
After mRNA encapsulation, purify and remove unencapsulated/loaded mRNA, free polymers or lipid materials, and adjust the final complex concentration, replace solvent buffer system, adjust pH value, etc. Through tangential flow filtration technology, mRNA LNP can be fully intercepted, while impurities can be washed and replaced with solvents. At the same time, Hzymes has established a series of mRNA LNP quality control release standards to strictly control the optimization results of LNP encapsulation and purification processes.




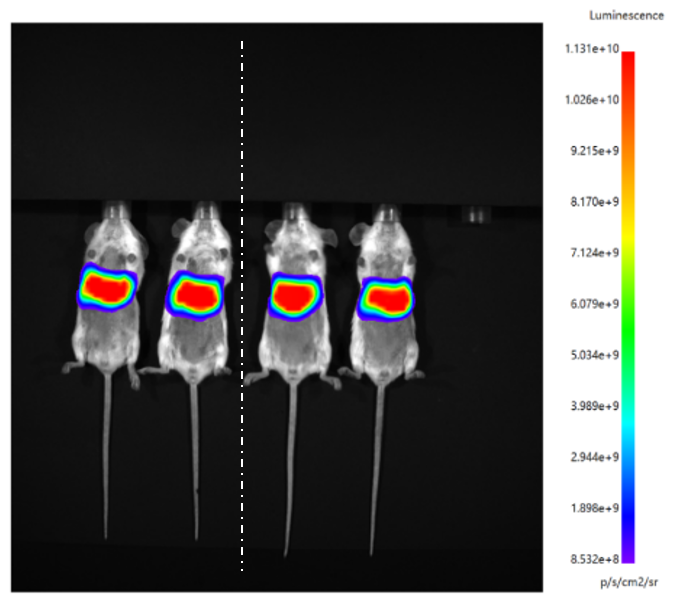

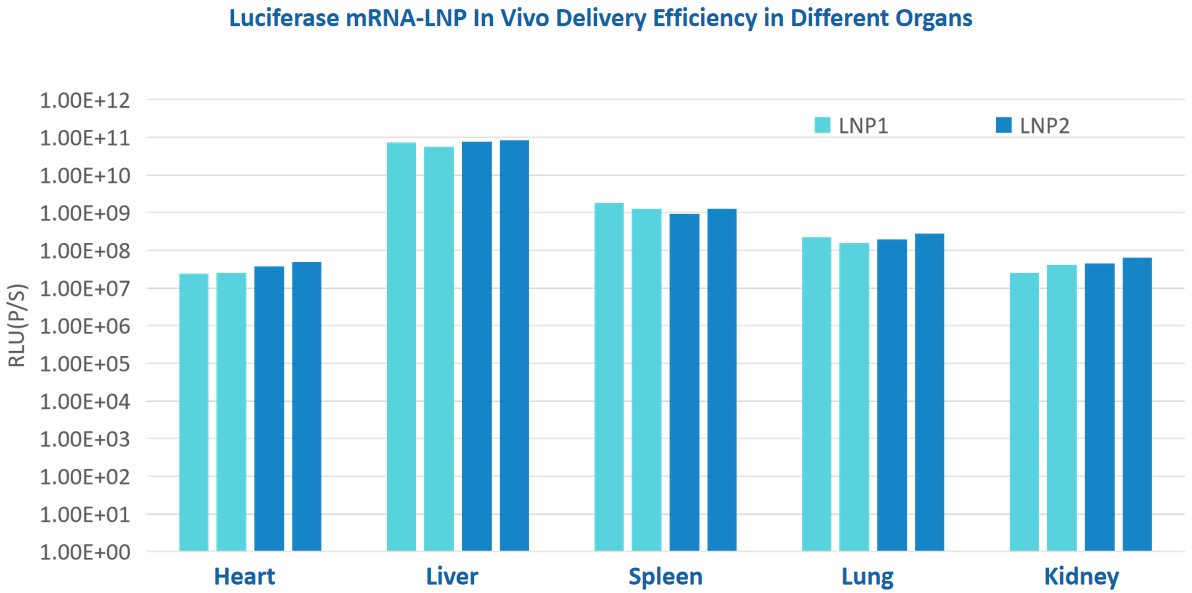
mRNA LNP quality inspection items
Quality inspection items | Quality inspection method |
| Entrapment efficiency | Ribogreen |
| Preparation RNA concentration | |
| PDI/particle size | DLS |
Zeta potentials | Doppler electrophoresis |
| Molecular molar ratio of multivalent RNA molecules | ddPCR+qPCR |
RNA sequence confirmation | Sanger sequencing/RT-PCR |
Determination of lipid content | ELSD-HPLC |
RNA integrity | CE/HPLC |
Endotoxin | Recombinant factor C/horseshoe crab reagent |
Protein expression validation | WB/FACS/IF/ELISA |


